Tβ4 (TB500) demonstrates measurable effects across cardiovascular, neurological, and cellular repair pathways through its fundamental role in actin regulation and cellular stress responses.
Laboratory studies reveal a distinctive dual-phase activity pattern where immediate cellular protection transitions into longer-term regenerative responses, establishing Tβ4’s research value from basic cell biology to complex tissue engineering applications with interconnected mechanisms suitable for multi-target experimental designs.
Cardiovascular and Cardiac Protection
Tβ4 demonstrates robust cardioprotective mechanisms through multiple pathways. The peptide enhances cardiac progenitor cell activation by stimulating epicardial progenitor cells and upregulating developmental factors like VEGF, Flk-1, TGF-β, FGFR-2, FGFR-4, FGF-17, and β-Catenin[1]. It directly modulates ROCK1 expression to reduce pathological scarring and fibroblast-to-myofibroblast transformation[2].
The peptide operates through a bi-phasic mechanism – an acute phase providing antiapoptotic and anti-inflammatory protection immediately post-injury, and a chronic phase activating vascular and cardiac progenitor cell growth[3]. Tβ4 protects cardiomyocytes from oxidative stress by targeting anti-oxidative enzymes and enhancing expression of antioxidant genes while reducing pro-apoptotic proteins[4].
Studies demonstrate that Tβ4 administration in experimental models significantly reduces infarct size, improves contractile performance, and prevents cardiac rupture following myocardial infarction[5]. The peptide enhances coronary vessel development and promotes neovascularization through activation of HIF-1α and p-HIF-1α via the AKT pathway[6].
Wound Healing and Tissue Repair
Tβ4’s wound healing properties center on its ability to promote angiogenesis, accelerate epithelialization, and modulate inflammatory responses[7]. The peptide enhances cell migration through its actin-sequestering properties and activates multiple wound healing pathways simultaneously[8].
In diabetic wound healing models, Tβ4 promotes VEGF/AKT pathway activation and increases CD31-positive endothelial cells, leading to improved vascularization[9]. The peptide demonstrates particular efficacy when delivered via controlled-release systems in research models, providing sustained biological effects over extended periods.
Anti-inflammatory Properties
Tβ4 exhibits potent anti-inflammatory mechanisms by inhibiting TNF-α-induced NF-κB activation and reducing IL-8 expression[10]. It enhances the production of specialized pro-resolving mediators (SPMs) through activation of 5-LOX and 12/15-LOX pathways, facilitating inflammation resolution[11].
Neurological and Neuroprotective Functions
Tβ4 provides comprehensive neuroprotection through multiple pathways. It promotes oligodendrocyte differentiation by upregulating p38 MAPK signaling and enhancing myelin basic protein expression[12]. The peptide suppresses TLR proinflammatory pathways by upregulating microRNA-146a, which inhibits IRAK1 and TRAF6[13].
In traumatic brain injury models, Tβ4 demonstrates both neuroprotective and neurorestorative effects, promoting neurogenesis, angiogenesis, and axonal remodeling when administered up to 6 hours post-injury[14]. The peptide enhances blood-brain barrier integrity by protecting tight junction proteins and reducing neuroinflammation[15].
Alzheimer’s Disease Research
Recent research reveals Tβ4’s ability to reverse glial cell phenotypic polarization and improve cognitive function in Alzheimer’s disease models through negative regulation of NF-κB signaling[16]. This mechanism involves modulating microglial activation states and reducing neuroinflammatory responses.
Antimicrobial and Immune Modulation
While Tβ4 lacks direct bactericidal activity, it demonstrates significant indirect antimicrobial effects by upregulating antimicrobial peptide production and enhancing bacterial killing when combined with antibiotics like ciprofloxacin[17]. The peptide modulates neutrophil function, reducing excessive inflammatory responses while maintaining effective pathogen clearance[18].
Macrophage Modulation
Tβ4 significantly influences macrophage effector function, downregulating reactive nitrogen species production and modulating efferocytosis activity[19]. This results in improved host defense responses while reducing tissue-damaging inflammatory cascades.
Metabolic and Cellular Regulation
A groundbreaking discovery reveals Tβ4’s role as an endogenous iron chelator and molecular switch for ferroptosis regulation[20]. This mechanism provides new insights into Tβ4’s cytoprotective properties and potential research applications in iron-related disorder studies.
Autophagy Regulation
Tβ4 demonstrates the ability to induce autophagy through inhibition of the PI3K/AKT/mTOR pathway, contributing to cellular homeostasis and stress response[21]. This mechanism is particularly relevant in hepatic stellate cell regulation and liver fibrosis prevention.
Mitochondrial Function
While Tβ4 shows variable effects on mitochondrial function across different cell types, it facilitates mitochondrial transfer from adipose-derived stem cells via tunneling nanotubes, enhancing cellular survival and regeneration[22].
Cancer Biology and Tumor Mechanisms
Tβ4 exhibits complex, context-dependent roles in cancer biology. In some cancers, overexpression correlates with metastasis and poor prognosis, as seen in thyroid cancer where increased Tβ4 expression associates with BRAF V600E mutations and advanced disease characteristics[23].
In diffuse-type gastric cancer, Tβ4 promotes metastasis through enhanced cell migration and anoikis resistance[24]. However, in multiple myeloma, decreased Tβ4 expression correlates with poor prognosis, suggesting tumor suppressive effects in certain malignancies[25].
Paradoxically, exogenous Tβ4 shows anti-cancer properties in lung cancer models, inhibiting tumor growth through JAK2/STAT3 pathway suppression. This suggests that endogenous versus exogenous Tβ4 may have different functional roles in cancer progression[26].
Regenerative and Anti-Aging Applications
Tβ4’s regenerative potential lies in its ability to reactivate embryonic developmental programs in adult tissues[27]. This mechanism involves stimulating dormant progenitor cell populations and enhancing their differentiation capacity across multiple organ systems.
The peptide significantly improves stem cell function by enhancing viability, reducing senescence, and promoting angiogenic factor secretion[28]. In diabetic endothelial progenitor cells, Tβ4 restores migration and tubule formation capabilities and functional capacity[29].
Research suggests Tβ4 could serve as a foundation for investigating regenerative mechanisms by utilizing developmentally essential molecules to study how adult organs might recapture embryonic regenerative capacity. This approach represents a paradigm shift from cell-based to factor-based regenerative research models.
Ocular Research
Tβ4 shows research promise in corneal wound healing studies and as an investigational compound for bacterial keratitis research models, where it enhances antibiotic efficacy while promoting tissue repair in laboratory settings[30]. The peptide’s anti-inflammatory and wound healing properties make it particularly suitable for ocular applications.
Pulmonary Applications
In lung pathology research models, Tβ4 demonstrates anti-fibrotic effects through TGF-β1 pathway regulation, suppressing fibroblast proliferation and epithelial-mesenchymal transition[31]. Nebulized delivery shows particular research interest for investigating idiopathic pulmonary fibrosis mechanisms.
Renal Protection
Tβ4 provides podocyte protection in kidney disease models, preventing cytoskeletal disorganization and maintaining glomerular filtration barrier integrity. This mechanism involves direct interaction with the actin cytoskeleton and anti-inflammatory effects[32].
This information is provided for research purposes only and is not intended to diagnose, treat, cure, or prevent any disease.
References
[1] I. Bock-Marquette et al., “Thymosin β4 mediated PKC activation is essential to initiate the embryonic coronary developmental program and epicardial progenitor cell activation in adult mice in vivo,” Elsevier BV, May 2009. doi: 10.1016/j.yjmcc.2009.01.017. Available: https://doi.org/10.1016/j.yjmcc.2009.01.017
[2] K. Maar, J. E. Thatcher, E. Karpov, S. Rendeki, F. Gallyas, and I. Bock-Marquette, “Thymosin Beta-4 Modulates Cardiac Remodeling by Regulating ROCK1 Expression in Adult Mammals,” MDPI AG, Apr. 2025. doi: 10.3390/ijms26094131. Available: https://doi.org/10.3390/ijms26094131
[3] W. Bao et al., “Cardioprotection by systemic dosing of thymosin beta four following ischemic myocardial injury,” Frontiers Media SA, 2013. doi: 10.3389/fphar.2013.00149. Available: https://doi.org/10.3389/fphar.2013.00149
[4] C. Wei, S. Kumar, I.-K. Kim, and S. Gupta, “Thymosin Beta 4 Protects Cardiomyocytes from Oxidative Stress by Targeting Anti-Oxidative Enzymes and Anti-Apoptotic Genes,” Public Library of Science (PLoS), Aug. 2012. doi: 10.1371/journal.pone.0042586. Available: https://doi.org/10.1371/journal.pone.0042586
[5] H. Peng et al., “Thymosin-β4prevents cardiac rupture and improves cardiac function in mice with myocardial infarction,” American Physiological Society, Sep. 2014. doi: 10.1152/ajpheart.00129.2014. Available: https://doi.org/10.1152/ajpheart.00129.2014
[6] S. Tang et al., “TMSB4 Overexpression Enhances the Potency of Marrow Mesenchymal Stromal Cells for Myocardial Repair,” Frontiers Media SA, Jun. 2021. doi: 10.3389/fcell.2021.670913. Available: https://doi.org/10.3389/fcell.2021.670913
[7] N. N. Raheem, A. S. Salman, and M. H. Jawad, “Histological Evaluation of Effect of Thymosin Beta 4 on Wound Healing of Skin,” European Open Science Publishing, May 2024. doi: 10.24018/ejdent.2024.5.3.330. Available: https://doi.org/10.24018/ejdent.2024.5.3.330
[8] G. Sosne, P. Qiu, and M. Kurpakus‐Wheater, “Thymosin beta 4: A novel corneal wound healing and anti-inflammatory agent,” Clinical Ophthalmology, vol. 1, pp. 201–207, 2007.
[9] D. Ti et al., “Controlled Release of Thymosin Beta 4 Using a Collagen–Chitosan Sponge Scaffold Augments Cutaneous Wound Healing and Increases Angiogenesis in Diabetic Rats with Hindlimb Ischemia,” Mary Ann Liebert Inc, Feb. 2015. doi: 10.1089/ten.tea.2013.0750. Available: https://doi.org/10.1089/ten.tea.2013.0750
[10] P. Qiu, M. K. Wheater, Y. Qiu, and G. Sosne, “Thymosin β4inhibits TNF‐α‐induced NF‐κB activation, IL‐8 expression, and the sensitizing effects by its partners PINCH‐1 and ILK,” Wiley, Feb. 2011. doi: 10.1096/fj.10-167940. Available: https://doi.org/10.1096/fj.10-167940
[11] Y. Wang, L. Banga, A. S. Ebrahim, T. W. Carion, G. Sosne, and E. A. Berger, “Activation of pro-resolving pathways mediate the therapeutic effects of thymosin beta-4 during Pseudomonas aeruginosa-induced keratitis,” Frontiers Media SA, Sep. 2024. doi: 10.3389/fimmu.2024.1458684. Available: https://doi.org/10.3389/fimmu.2024.1458684
[12] M. Santra et al., “Thymosin beta 4 mediates oligodendrocyte differentiation by upregulating p38 MAPK,” Wiley, Aug. 2012. doi: 10.1002/glia.22400. Available: https://doi.org/10.1002/glia.22400
[13] M. Santra et al., “Thymosin β4 Up-regulation of MicroRNA-146a Promotes Oligodendrocyte Differentiation and Suppression of the Toll-like Proinflammatory Pathway,” Elsevier BV, Jul. 2014. doi: 10.1074/jbc.m113.529966. Available: https://doi.org/10.1074/jbc.m113.529966
[14] Y. Xiong et al., “Neuroprotective and neurorestorative effects of thymosin β4 treatment following experimental traumatic brain injury,” Wiley, Oct. 2012. doi: 10.1111/j.1749-6632.2012.06683.x. Available: https://doi.org/10.1111/j.1749-6632.2012.06683.x
[15] W. Stewart, C. Hejl, R. Guleria, and S. Gupta, “Effect of thymosin β4 on lipopolysaccharide‑stimulated brain microvascular endothelial cell remodeling: A possible role in blood‑brain barrier injury,” Spandidos Publications, Aug. 2023. doi: 10.3892/etm.2023.12167. Available: https://doi.org/10.3892/etm.2023.12167
[16] M. Wang et al., “Thymosin β4 reverses phenotypic polarization of glial cells and cognitive impairment via negative regulation of NF-κB signaling axis in APP/PS1 mice,” Springer Science and Business Media LLC, Jun. 2021. doi: 10.1186/s12974-021-02166-3. Available: https://doi.org/10.1186/s12974-021-02166-3
[17] T. W. Carion et al., “Antimicrobial Effects of Thymosin Beta-4 and Ciprofloxacin Adjunctive Therapy in Pseudomonas aeruginosa Induced Keratitis,” MDPI AG, Sep. 2020. doi: 10.3390/ijms21186840. Available: https://doi.org/10.3390/ijms21186840
[18] Y. Wang, T. W. Carion, A. S. Ebrahim, G. Sosne, and E. A. Berger, “Adjunctive Thymosin Beta-4 Treatment Influences PMN Effector Cell Function during Pseudomonas aeruginosa-Induced Corneal Infection,” MDPI AG, Dec. 2021. doi: 10.3390/cells10123579. Available: https://doi.org/10.3390/cells10123579
[19] M. Kasprzak, H. Lærke, F. Larsen, K. Knudsen, S. Pedersen, and A. Jørgensen, “Kasprzak, M.M., et al., Correction: Effect of Enzymatic Treatment of Different Starch Sources on the in Vitro Rate and Extent of Starch Digestion. Int. J. Mol. Sci. 2012, 13, 929-942.,” MDPI AG, Dec. 2012. doi: 10.3390/ijms131217292. Available: https://doi.org/10.3390/ijms131217292
[20] J. I. Lachowicz et al., “Thymosin β4 Is an Endogenous Iron Chelator and Molecular Switcher of Ferroptosis,” MDPI AG, Jan. 2022. doi: 10.3390/ijms23010551. Available: https://doi.org/10.3390/ijms23010551
[21] J. Choi et al., “Thymosin Beta 4 Inhibits LPS and ATP-Induced Hepatic Stellate Cells via the Regulation of Multiple Signaling Pathways,” MDPI AG, Feb. 2023. doi: 10.3390/ijms24043439. Available: https://doi.org/10.3390/ijms24043439
[22] Y. Ying, N. Tao, F. Zhang, X. Wen, M. Zhou, and J. Gao, “Thymosin β4 Regulates the Differentiation of Thymocytes by Controlling the Cytoskeletal Rearrangement and Mitochondrial Transfer of Thymus Epithelial Cells,” MDPI AG, Jan. 2024. doi: 10.3390/ijms25021088. Available: https://doi.org/10.3390/ijms25021088
[23] C.-Y. Kuo, J.-Y. Jhuang, W.-C. Huang, and S.-P. Cheng, “Aberrant Expression of Thymosin Beta-4 Correlates With Advanced Disease and BRAF V600E Mutation in Thyroid Cancer,” SAGE Publications, Oct. 2022. doi: 10.1369/00221554221138370. Available: https://doi.org/10.1369/00221554221138370
[24] H. An et al., “In vivo CRISPR–Cas9 knockout screening using quantitative PCR identifies thymosin beta‐4 X‐linked that promotes diffuse‐type gastric cancer metastasis,” Wiley, Jun. 2021. doi: 10.1002/mc.23326. Available: https://doi.org/10.1002/mc.23326
[25] J. Caers et al., “Thymosin 4 has tumor suppressive effects and its decreased expression results in poor prognosis and decreased survival in multiple myeloma,” Ferrata Storti Foundation (Haematologica), Oct. 2009. doi: 10.3324/haematol.2009.006411. Available: https://doi.org/10.3324/haematol.2009.006411
[26] R. Yu et al., “Exogenous Thymosin Beta 4 Suppresses IPF-Lung Cancer in Mice: Possibly Associated with Its Inhibitory Effect on the JAK2/STAT3 Signaling Pathway,” MDPI AG, Feb. 2023. doi: 10.3390/ijms24043818. Available: https://doi.org/10.3390/ijms24043818
[27] K. Maar et al., “Utilizing Developmentally Essential Secreted Peptides Such as Thymosin Beta-4 to Remind the Adult Organs of Their Embryonic State—New Directions in Anti-Aging Regenerative Therapies,” MDPI AG, May 2021. doi: 10.3390/cells10061343. Available: https://doi.org/10.3390/cells10061343
[28] L. Su et al., “Thymosin beta-4 improves endothelial function and reparative potency of diabetic endothelial cells differentiated from patient induced pluripotent stem cells,” Springer Science and Business Media LLC, Jan. 2022. doi: 10.1186/s13287-021-02687-x. Available: https://doi.org/10.1186/s13287-021-02687-x
[29] Z. Quan, Q.-L. Wang, P. Zhou, G.-D. Wang, Y.-Z. Tan, and H.-J. Wang, “Thymosin β4 promotes the survival and angiogenesis of transplanted endothelial progenitor cells in the infarcted myocardium,” Spandidos Publications, Apr. 2017. doi: 10.3892/ijmm.2017.2950. Available: https://doi.org/10.3892/ijmm.2017.2950
[30] G. Sosne and E. A. Berger, “Thymosin beta 4: A potential novel adjunct treatment for bacterial keratitis,” Elsevier BV, May 2023. doi: 10.1016/j.intimp.2023.109953. Available: https://doi.org/10.1016/j.intimp.2023.109953
[31] R. Yu et al., “Inhaled exogenous thymosin beta 4 suppresses bleomycin-induced pulmonary fibrosis in mice via TGF-β1 signalling pathway,” Oxford University Press (OUP), Nov. 2024. doi: 10.1093/jpp/rgae143. Available: https://doi.org/10.1093/jpp/rgae143
[32] W. Mason, “The role of thymosin beta 4 in renal podocyte function,” 2021.



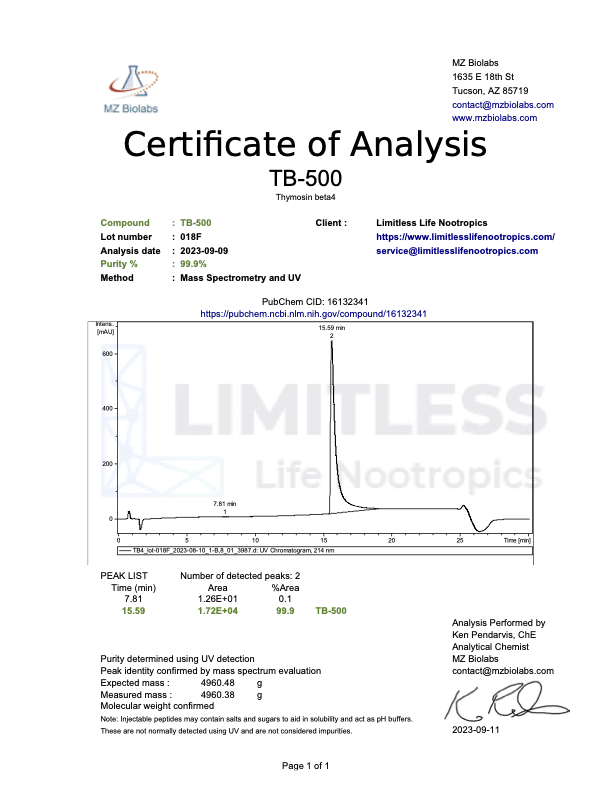
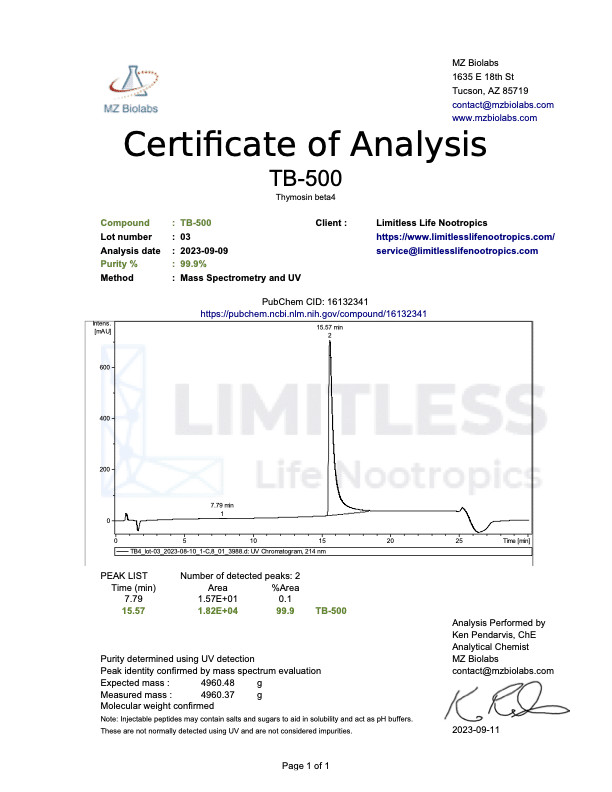
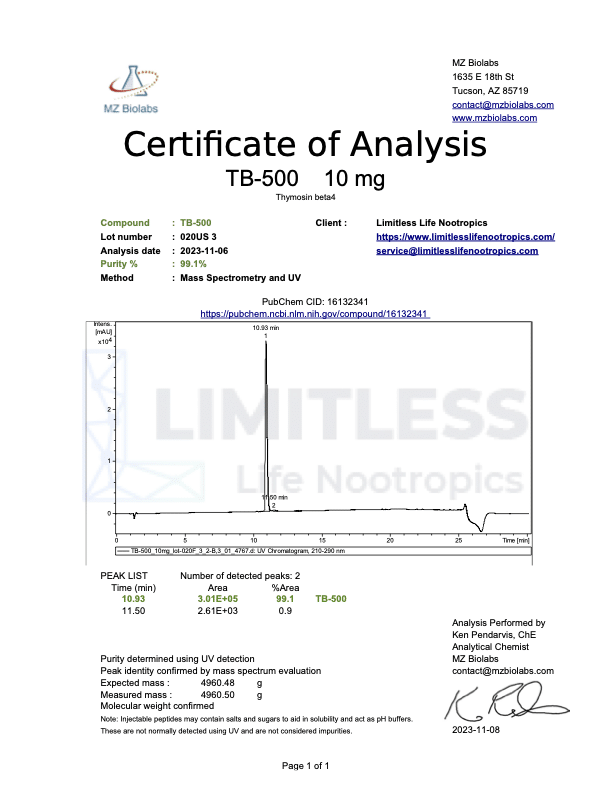
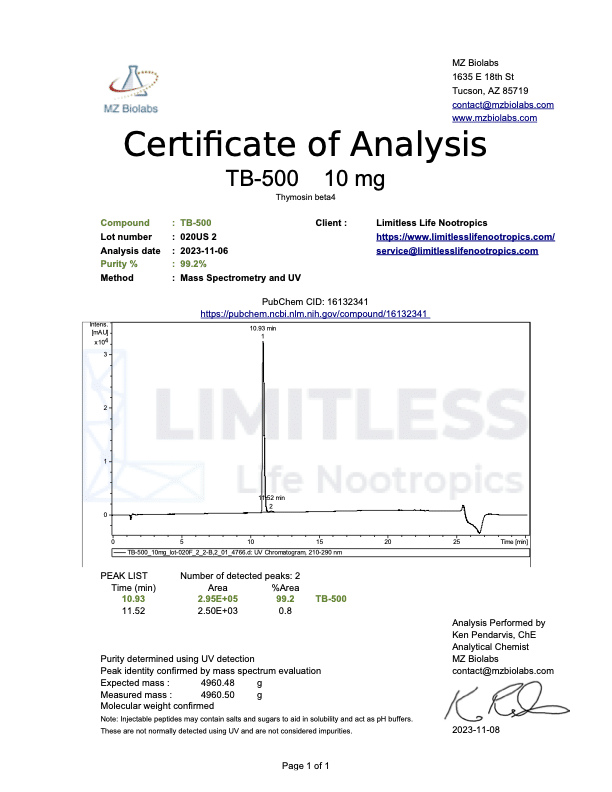













 <img class="wp-image-136359 aligncenter entered exited" src="data:;base64,” alt=”COA_TB-500_US 1146_r2_2024-07-09″ width=”452″ height=”640″ data-lazy-srcset=”https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-212×300.png 212w, https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-724×1024.png 724w, https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-768×1087.png 768w, https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-600×849.png 600w, https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m.png 950w” data-lazy-sizes=”(max-width: 452px) 100vw, 452px” data-lazy-src=”https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-212×300.png” />
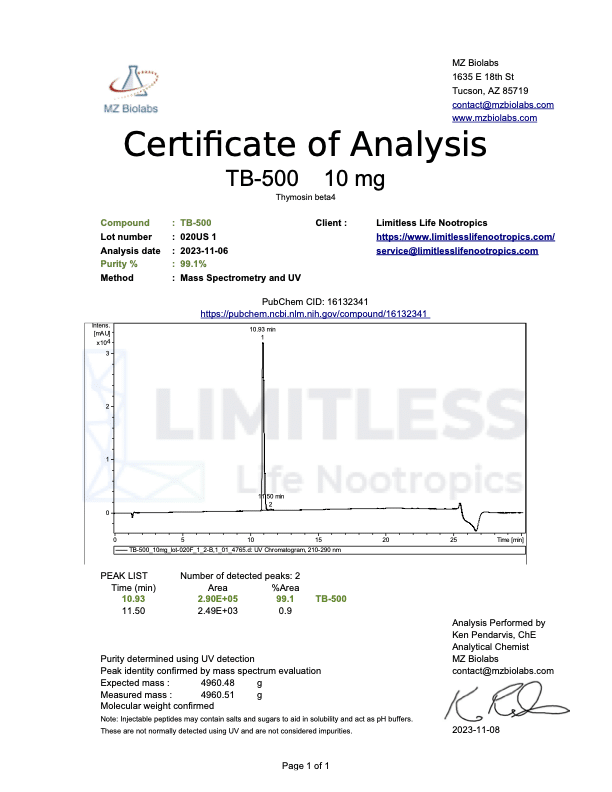
<img class="wp-image-136359 aligncenter entered exited" src="data:;base64,” alt=”COA_TB-500_US 1146_r2_2024-07-09″ width=”452″ height=”640″ data-lazy-srcset=”https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-212×300.png 212w, https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-724×1024.png 724w, https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-768×1087.png 768w, https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-600×849.png 600w, https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m.png 950w” data-lazy-sizes=”(max-width: 452px) 100vw, 452px” data-lazy-src=”https://www.limitlesslifenootropics.com/wp-content/uploads/2019/08/Captura-de-pantalla-2024-07-10-a-las-10.45.58 a.-m-212×300.png” />































































Reviews
There are no reviews yet.